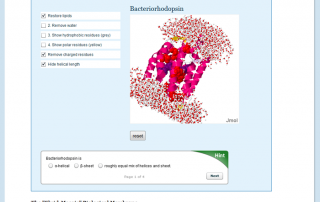
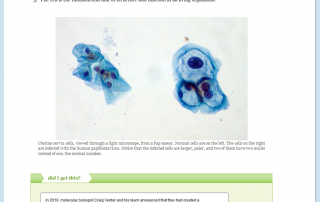
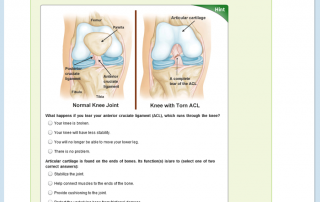
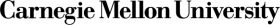Chemistry Study Skills: The Student Cognition Toolbox — Open & Free
The Student Cognition Toolbox (SCT) is a short course companion (or “sidecar”) introducing and providing direct practice in study strategies that are informed by learning science research. In this version of the SCT, material from introductory chemistry is used to illustrate the application of the study strategies. This version draws from material in CMU OLI’s